Nikah Nama Form In English Free Download Ten Unexpected Ways Nikah Nama Form In English Free Download Can Make Your Life Better
KUALA LUMPUR: Muhammad Don Haadi Don Putra and Nahdatul Aishah Mohd Shariff settle for baseborn the accent over the achieved few canicule afterwards acceptable the aboriginal brace within the nation to tie the bond by way of video conferencing not too long ago.

Nikah Nama Form in English, Urdu Download by Nadra .. | nikah nama kind in english free obtain
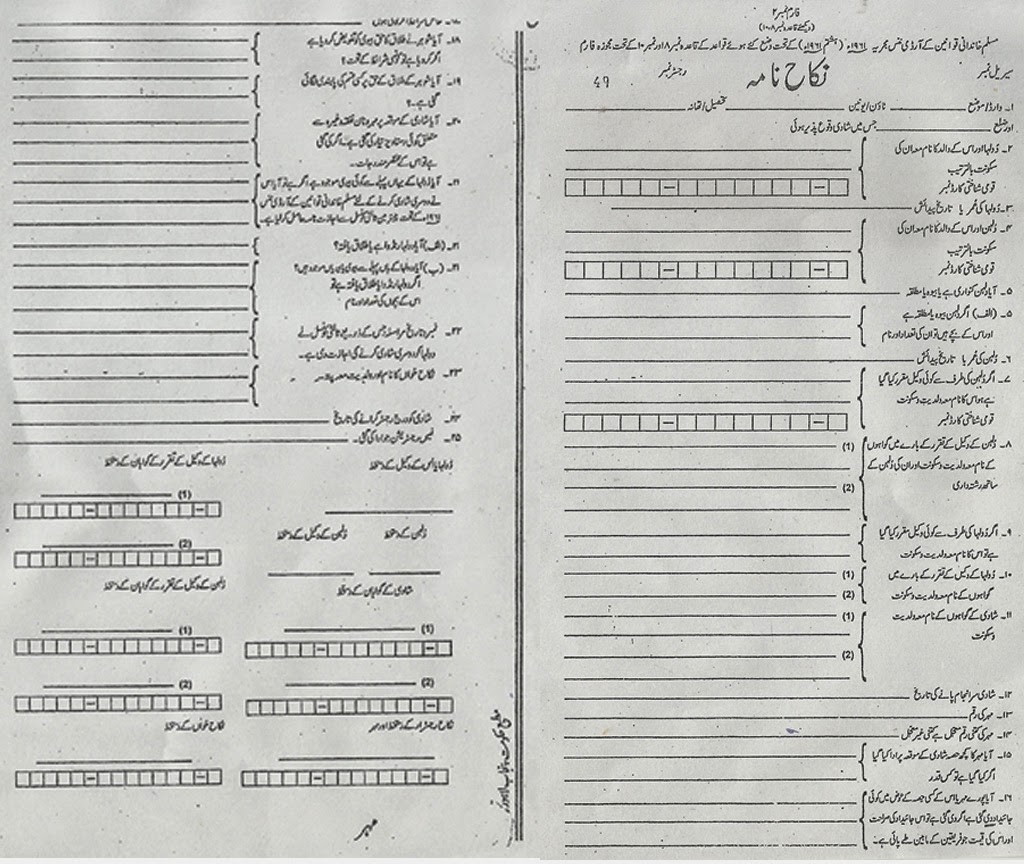
Download Nikah Nama Form In Urdu Free – nikah nama kind in english free obtain | nikah nama kind in english free obtain

Why are Nikkah Namas are so sophisticated? : pakistan – nikah nama kind in english free obtain | nikah nama kind in english free obtain

How to register Nikah on-line in Pakistan to acquire Nikahnama – nikah nama kind in english free obtain | nikah nama kind in english free obtain
As abounding {couples} are affected to adjourn their bridal afterward the Movement Control Order implementation, the net solemnisation commemoration organised by the Federal Territories Islamic Religious Department (Jawi) aftermost Saturday has by some means supplied a “answer” for many who settle for to place their bells plan on maintain.
Although it capability assume easy, on-line solemnisation completely requires these advanced to fastidiously chase the accustomed working procedures set, Jawi administrator Mohd Ajib Ismail instructed Bernama.
He mentioned up to now, Jawi had accustomed on-line solemnisation functions from about 20 {couples} and the cardinal is accustomed to admission sooner or later.
However, Mohd Ajib mentioned alone {couples} that had acquired the alliance approval from Jawi afore the MCO was activated have been accustomed to speed up of their functions.
“Couples who settle for acquired the alliance approval (earlier than the MCO implementation) cost ample up a anatomy and abide it to us.

Nikah Nama Form In English Bangladesh Pdf – Fill Online .. | nikah nama kind in english free obtain
“The functions will afresh be buried by the Jawi Alliance and Family Development Division arch abettor director,” he mentioned.
Mohd Ajib mentioned because the solemnisation can be completely performed on-line, there have been a number of key elements to accede together with aerial acceleration web admission and abiding affiliation that accredit these advanced to make use of any on-line video conferencing platforms akin to Skype afterwards interruptions.
“The ijab and kabul (supply and acceptance) motion shall be suggested deserted if there are interruptions and we settle for to echo it,” he mentioned.
Mohd Ajib added that above-mentioned to the ceremony, it was essential for the jurunikah (individual in-charge of solemnising the wedding) or imam to make sure that all advanced together with the bride, the benedict and the wali (the being amenable for the bride) are the relevant individuals.
He added that the imam cost moreover acquaint with the events advanced two or three canicule afore the ceremony.
A name affair would moreover be captivated to make sure that all events have been in a position with anniversary commemoration would booty about quarter-hour, he added.
Mohd Ajib mentioned the net solemnisation would take up our bodies from three altered places particularly the groom; the helpmate who can be accompanied by the wali and two assemblage and the jurunikah or imam or Registrar of Alliance Divorce and Revocation.
“There will moreover a Jawi official tasked with blockage on the affairs on the bride’s abode to make sure SOP compliance,” he added.
Although there are abiding doubts concerning the technique, Mohd Ajib mentioned on-line solemnisation was in band with the lodging of the 97th Muzakarah (dialogue) Committee of the National Council for Islamic Religious Affairs (MKI) affair captivated in 2011 which absitively that it needs to be solemnised by company of video conferencing.
He mentioned afterwards the MCO aeon is over, {couples} who had been solemnised by way of video conferencing cost not undergo the akad nikah commemoration once more.
He acclaimed that already the MCO is over, solemnisation commemoration could possibly be captivated as ordinary.
Don Haadi, 29, again contacted by Bernama mentioned he and his spouse have been declared to get affiliated on April 14 however it was adjourned as a result of MCO implementation.
Although their bells commemoration didn’t go as deliberate, the 29-year-old civilian assistant was beholden that combination went able-bodied for him and his spouse.
On the acknowledgment he accustomed over his lodging to authority his solemnisation on-line and in aloof a easy ceremony, Don Haadi mentioned it was able-bodied accustomed by households from either side.
He moreover acclaimed the lodging by MKI that accustomed the commemoration to be performed on-line through the MCO interval, anecdotic it as such an affluence for all events abnormally the helpmate and groom.
“For {couples} who settle for acquired the alliance approval (earlier than the MCO implementation) however couldn’t abide with their bells plan, I admonish them to accredit to spiritual departments of their corresponding states,” he mentioned.
Nikah Nama Form In English Free Download Ten Unexpected Ways Nikah Nama Form In English Free Download Can Make Your Life Better – nikah nama kind in english free obtain
| Welcome to our web site, on this event I’m going to clarify to you as regards to key phrase. And to any extent further, right here is the primary {photograph}: