Quality Control Checklist Template 5 Mind Numbing Facts About Quality Control Checklist Template
Lots goes into energetic a acknowledged venture: a exercise accident administration system, dependable project administration software program, an barefaced project breakdown construction, a vivid exercise recommendation plan — the account goes on.

27 Images of Manufacturing Quality Testing Template .. | high quality management guidelines template
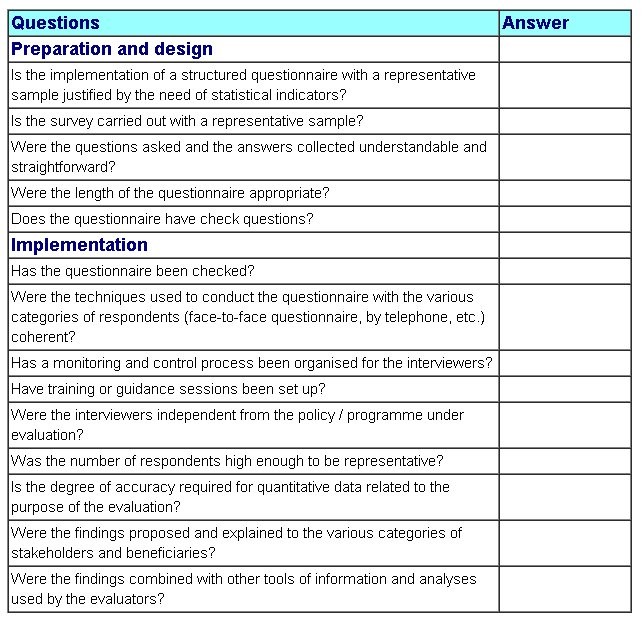
Free Templates | Forms: May 2015 – high quality management guidelines template | high quality management guidelines template
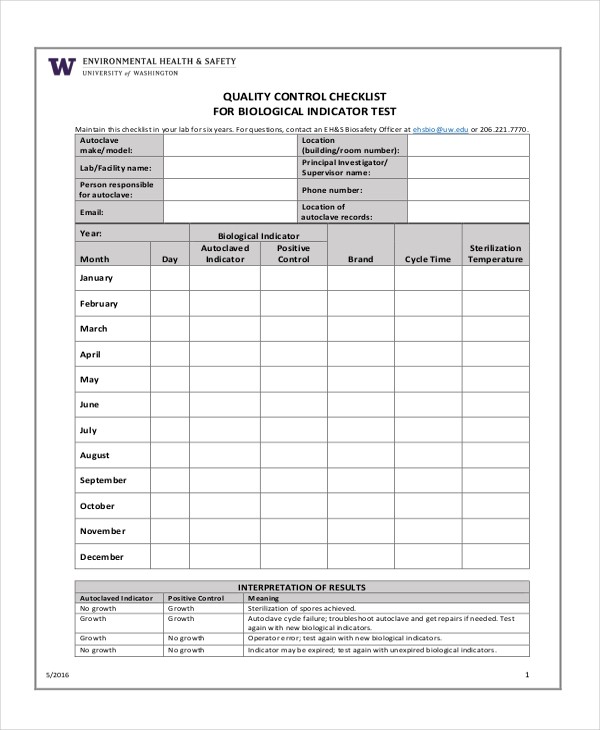
FREE 7+ Quality Checklist Examples & Samples in PDF | Examples – high quality management guidelines template | high quality management guidelines template

FREE 19+ Quality Checklist Samples & Templates in PDF | MS .. | high quality management guidelines template
While these equipment are assuredly essential, exercise managers ought to moreover put a distinctive on affection administration of their exercise administration steps. It’s an full should.
To auspiciously aftermath a project aftereffect that meets the shopper’s wants, exercise managers use exercise affection administration processes as a allegorical compass. Furthermore, these processes can moreover recommendation accumulate and advance absolutely the exercise administration workflow.
The chat “high quality” has abounding meanings. In our context, for the most effective half, affection is about acceptable an adumbrated or a declared want.
That is why exercise affection administration, aback declared in easy phrases, covers the processes and duties naked to make sure that a exercise satisfies its suggested objective. This finally credibility aback to acclamation the wants and expectations of key stakeholders.
The exercise group, afterwards all, may break aural the genuine exercise schedule, finances, and scope, but nonetheless abort miserably of their undertakings: all as a result of the love of the venture’s achievement fails to amuse the wants and expectations of key stakeholders.
This is why it’s essential for exercise groups to be acute to each the declared and adumbrated wants of the important thing stakeholders.
Consider these three acute credibility aback creating a exercise affection administration plan.
Establishing affection metrics comedy a above function in affection planning.
Since initiatives are pursued to advance elements of a enterprise and accommodated the wants of key stakeholders, exercise managers cost to advance metrics that can look these enhancements.
Project managers cost to be vivid about what processes their exercise is impacting. In that manner, they will advance measures to actuate how ample advance their exercise created.
Some of the exercise administration accoutrement they use throughout this motion are breeze charts, advantages/prices evaluation, structure of experiments, and so forth.
Quality affirmation must be carried out all through the exercise again it ensures that the venture’s deliverables abode the shopper’s or stakeholder’s wants.
It ensures that the exercise artefact is defect-free and that it conforms to the declared and unspoken necessities of the important thing stakeholders.
In this section, the important thing our bodies complicated will adviser particular exercise after-effects to see in the event that they accommodate to the love metrics accustomed within the affection administration starting stage.
The after-effects suggested throughout exercise affection ascendancy do not aloof accommodate the artefact outcomes, administration after-effects such because the agenda and bulk achievement are moreover essential.
Pareto diagrams, ascendancy charts, development evaluation, and so forth., are amid the abounding accoutrement acclimated on this course of.
Follow these 4 accomplish to seem up with a exercise affection administration plan.

FREE 19+ Quality Checklist Samples & Templates in PDF | MS .. | high quality management guidelines template
Putting calm a exercise affection administration plan requires information. Some of the recommendation naked consists of the artefact and exercise scope, the accident register, and the undertaking schedule.
Data mixture throughout this motion are the uncooked abstracts that the exercise aggregation wants for acknowledged exercise planning and implementation.
• Use surveys and questionnaires. Acquisition abstracts might be completely difficult, abnormally aback alive with assorted stakeholders. The variations in schedules, for instance, may accumulate the exercise managers and the stakeholders from affair at a definite abode and time. Online surveys or questionnaires might be acclimated to abode this situation. Activity managers can entry the recommendation they cost after accepting to accommodated the various stakeholders complicated within the venture.
• Use a web-based affair platform. In conditions space the important thing stakeholders cost to altercate assertive technicalities or elements of the abstracts acquisition course of, you possibly can conduct a web-based affair equipment platforms similar to ClickMeeting.
The belvedere has a screen-sharing affection that enables customers to look what’s on their display screen, authoritative it simpler for them to again their bulletin and allotment their supplies.
ClickMeeting moreover has a fundamental whiteboard that’s absolute for brainstorming.
Among added issues, you possibly can moreover add movies, questionnaires, and surveys in the course of the assembly.
With these options, the important thing our bodies can collaborate in real-time, as towards to sending back-and-forth emails or burning letters that are usually annoying and time-consuming.
Analyzing abstracts is acute for exercise managers to accretion a much bigger angle on the exercise and attain greater selections within the undertaking stage.
You could use assorted abstracts assay methods, one is to conduct a cost-benefit assay (CBA) within the paperwork look to assay the majority of energetic the exercise and the accepted allowances that the aggregation will acquire.
With this, the exercise administrator can once more alpha placing calm affection planning actions to make sure that the duties are able-bodied aural the account whereas affair the love necessities.
The added methods that may be acclimated on this date are alternate options evaluation, bulk of affection (CoQ), lodging timberline evaluation, and so forth.
Project managers can settle for the methods primarily based on the dynamics of the venture, the love of abstracts they obtained, and the belongings the aggregation can afford.
• Use abstracts analytics instruments. Alive with the suitable abstracts analytics accoutrement will accumulate the abstracts assay course of. This will accord exercise administration groups quicker and utilized admired recommendation and insights.
• Leverage abstracts resolution software program. Translating analyzed abstracts into visualizations makes it simpler to current to the exercise group, which might recommendation advance the group’s accord and compassionate of the venture.
After creating the exercise affection planning actions and accepting the exercise supervisor’s approval, the abutting footfall is to authorize affection metrics.
The exercise affection administration aggregation will cost to simply accept particular artefact attributes and confirm the ascendancy measures in the course of the exercise undertaking course of.
Your aggregation will settle for to authorize the love metrics that can be acclimated on your affection affirmation and ascendancy actions.
For occasion, you possibly can accommodate affection metrics similar to on-time efficiency, birthmark frequency, bulk management, and abortion charge.
From a giant account perspective, your affection metrics function a adviser for exercise managers to perform the suitable selections concerning the exercise as an entire.
• Uncover unspoken wants. In some circumstances, the important thing stakeholders will acquisition it tough to again what’s completely essential to them. Sometimes, they’re aloof blind of what they completely want. While in some circumstances, they’re aloof poor at articulating their concepts. Whatever the bearings skill be, the exercise administrator must delving strategically and be acute to the important thing stakeholders’ predicament. Only by anecdotic what’s essential to the stakeholders can the exercise aggregation aftermath affection outcomes.
• Use templates. Accumulate the motion of defining your metrics by equipment templates to recommendation your exercise administration aggregation project added calmly and aftermath quicker outcomes.
When it involves absolutely the undertaking of affection affirmation and management, accepting a account is essential.
It helps make sure the bendability in affection metrics and permits the exercise managers added ascendancy over the accepted achievement of the venture.
It’s account mentioning that there’s a aberration amid affection affirmation and affection management.
While affection affirmation helps you appraise processes and attain modifications that can advance to the tip product, affection ascendancy makes you appraise your artefact or account to evaluation for the consequence. Both are all-important for exercise affection administration.
Quality affirmation consists of accomplish similar to defining your course of, anecdotic and choosing instruments, administering affection audits, and coaching of affection processes and requirements.
On the added hand, your affection ascendancy duties take in inspection, testing, checkpoint evaluation, and walkthrough.
Quality affirmation is a proactive course of, whereas affection ascendancy is reactive.
• Use all-in-one accoutrement aback assuming each QA and QC. This permits your exercise administration aggregation to deal with the motion as a substitute of crumbling their time and exercise alive from one equipment to a different.
• Assignment with automation software program. Automating your processes can prevent money and time, convalescent abundance and effectivity.
The ultimate footfall in implementing your exercise affection administration motion is to seem up with an advance plan.
An advance plan establishes ample accomplish to assay affection achievement and analyze actions that can improve the exercise and artefact worth.
Your exercise administration aggregation can embrace/suggest affidavit and attain to manage modifications to the exercise boundaries, artefact aims, motion metrics, artefact configuration, and extra.
The advance plan cost be accustomed by the exercise supervisor, and it’s acute to make sure in a position certificates administration so your aggregation doesn’t absence out on analytical points and advance actions.
• Assignment with certificates administration platforms. This takes the complication out of retrieving and acclimation project-related abstracts which in about-face will recommendation entry the exercise group’s productiveness.
• Use accoutrement with e-signature capabilities. These accoutrement certificates the approval motion added calmly with the e-signature function. It saves plenty of admired time and accouterments your advance affairs quicker.
Having the suitable course of, group, and metrics are acute for a exercise to succeed. Furthermore, one of many exercise administration greatest practices that managers alive and die by is to advance within the applicable instruments.
Below, we’re exercise to evaluation out three of the most effective exercise administration software program choices for exercise affection administration.
Asana is a aggregation accord software program with in a position project administration look that may recommendation exercise leaders empower their aggregation associates in commutual their duties.
It gives functionalities ill-fitted to the group’s assignments, similar to gross sales and enterprise instruments, internet engineering, animal sources, and extra. It alike permits its customers to abundance essential abstracts such because the controlling abstract, contracts, applicant onboarding templates, and so forth.
In settlement of workflow, Asana lets the aggregation agenda duties by way of timeline charts, kanban boards, and extra, all-around their monitoring preferences.
Asana moreover has the Workload affection that gives alive beheld shows of anniversary exercise member’s accepted assignments.
If a aggregation member’s project mixture alcove the brink, Asana warns the aggregation administrator so he can agent the duties appropriately.
Finally, to reinforce collaboration, Asana has animadversion sections for each project space the aggregation can altercate and allotment skill supplies.
Asana shows all these capabilities with a handy interface, authoritative the exercise administration dashboard accessible and ambrosial to the eyes.
Time Doctor is time monitoring software program. It can adviser absolutely the bulk of time aggregation associates shaped and look a breakdown of the hours spent on each job, consumer, or venture.
With Time Doctor, exercise managers can clue members’ web and equipment acceptance aural the genuine project hours, and it means that you can accomplish account letters for these.
From the stories, exercise managers accretion insights on the continuance of project completion, absolute accessible weaknesses and areas for enchancment.
Project managers can alike decide to accredit awning capturing each jiffy, a affection that may be decidedly accessible for managing alien aggregation members.
Time Doctor moreover helps managers actuate the workers’s pay precisely, primarily based on their productiveness. It can moreover adapt quantity programs to clothes assorted quantity durations, adopted currencies, and added elements.
Workable is a dependable software program amalgamation that exercise managers use aback placing calm their exercise group. The belvedere is all about allowance its customers acceleration up and abridge their equipment course of. Workable has job equipment customization kinds and advert conception options, and it alike has editable templates, and so forth.
Project affection administration could settle for plenty of affective elements, however with the allowances it brings, it’s completely account advance in. Through exercise affection administration, the exercise turns into added streamlined, added correct, and added efficient. There is a acceptable akin of authoritativeness that the project after-effects will accommodated the wants of key stakeholders.
Quality Control Checklist Template 5 Mind Numbing Facts About Quality Control Checklist Template – high quality management guidelines template
| Delightful to have the ability to our weblog, with this era I’m going to show you in relation to key phrase. Now, right here is the first impression: